Abstract
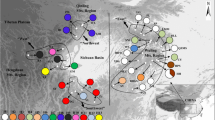
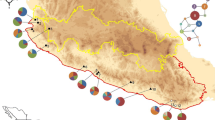
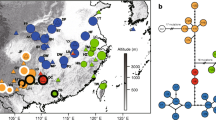
Limited data are available on genetic structures of the herbaceous plant species populations inhabiting mountainous areas of Siberia and Northeastern Asian (Russian Far East). Although this area was not directly impacted by the extensive ice-sheets during the Quaternary, it experienced significant climatic fluctuations that influenced rich local flora. Orostachys spinosa (Crassulaceae) lacks any adaptations for long-distance dispersal, yet the species is characterized by an unusually wide range spanning from the Urals to the coast of the Pacific Ocean. We studied O. spinosa phylogeography and genetic diversity across its range sampling 203 individuals from 21 natural populations. Using sequences from three chloroplast DNA non-coding regions, we revealed 82 haplotypes and observed high level of population differentiation indicating presence of the phylogeographic structure (GST = 0.501 and NST = 0.822 (p < 0.01)). In concordance with the previous phylogenetic analyses based on ITS rDNA data, parsimony network revealed two distinct cpDNA haplotype lineages deferring in their structure and characteristics of genetic diversity. The split between these haplotype groups can be dated to the Pliocene (ca. 3.6 Mya). According to our estimates diversification in the Western group of populations took place ca. 1 Mya earlier than in the Eastern group (3.1 Mya and 2.26 Mya, respectively). Apart from the generally accepted notion about East Asian origin of O. spinosa, our results indicated that the species could have originated in mountains of Southern Siberia (Altai). This region was colonized independently from O. thyrsiflora which has a largely overlapping distribution in this area.




Similar content being viewed by others
References
Abbott RJ, Smith LC, Milne RI, Crawford RM, Wolff K, Balfour J (2000) Molecular analysis of plant migration and refugia in the Arctic. Science 289:1343–1346. https://doi.org/10.1126/science.289.5483.1343
Allen GA, Marr KL, McCormick LJ, Hebda RJ (2012) The impact of Pleistocene climate change on an ancient arctic–alpine plant: multiple lineages of disparate history in Oxyria digyna. Ecol Evol 2:649–665. https://doi.org/10.1002/ece3.213
Ansell SW, Stenøien HK, Grundmann M, Russell SJ, Koch MA, Schneider H, Vogel JC (2011) The importance of Anatolian mountains as the cradle of global diversity in Arabis alpina, a key arctic–alpine species. Ann Bot (Oxford) 108:241–252. https://doi.org/10.1093/aob/mcr134
Avise JC (2000) Phylogeography: the history and formation of species. Harvard University Press, Camridge
Avise JC (2004) Molecular markers, natural history and evolution, 2nd edn. Sinauer, Sunderland
Bezdeleva TA (1995) Crassulaceae. In: Kharkevicz SS (ed) Plantae vasculares Orientis Extremi Sovietici, vol. 7. Nauka, Sankt-Petersburg, pp 214–235 (in Russian)
Blomdin R, Heyman J, Stroeven AP, Harbora JM, Gribenski N, Jansson KN, Petrakov DA, Ivanov MN, Orkhonselenge A, Rudoye AN, Walther M (2016) Glacial geomorphology of the Altai and western Sayan mountains, central Asia. J Maps 12:123–136. https://doi.org/10.1080/17445647.2014.992177
Blyakharchuk TA, Wright HE, Borodavko PS, van der Knaap WO, Ammann B (2007) Late glacial and Holocene vegetational history of the Altai mountains (southwestern Tuva Republic, Siberia). Palaeogeogr Palaeoclimatol Palaeoecol 245:518–534. https://doi.org/10.1016/j.palaeo.2006.09.010
Bonfield JK, Smith KF, Staden R (1995) A new DNA sequence assembly program. Nucl Acids Res 23:4992–4999. https://doi.org/10.1093/nar/23.24.4992
Byalt VV (1999) Monograph of the genus Orostachys Fisch. (Crassulaceae). Dissertation, Botanical Institute, Russian Academy of Sciences, Saint-Petersburg, p. 288 (in Russian)
Chen JM, Du ZY, Yuan YY, Wang QF (2014) Phylogeography of an alpine aquatic herb Ranunculus bungei (Ranunculaceae) on the Qinghai-Tibet Plateau. J Syst Evol 52:313–325. https://doi.org/10.1111/jse.12093
Chernykh DV, Zolotov DV, Yamskikh GY, Grenaderova AV (2014) Postglacial environmental change in the valley of Malye Chily River (the basin of Lake Teletskoye), northeastern Russian Altai. Phys Geogr 35:390–410. https://doi.org/10.1080/02723646.2014.929881
Chytrý M, Horsák M, Syrovátka V, Danihelka J, Ermakov N, German DA, Hájek M, Hájek O, Hájková P, Horsáková V, Kočí M, Kubešová S, Lustyk P, Nekola JC, Preislerová Z, Resl P, Valachovič M (2017) Refugial ecosystems in central Asia as indicators of biodiversity change during the Pleistocene-Holocene transition. Ecol Indicat 76:357–367. https://doi.org/10.1016/j.ecolind.2016.12.033
Clement M, Posada D, Crandall K (2000) TCS: a computer program to estimate gene genealogies. Molec Ecol 9:1657–1660. https://doi.org/10.1046/j.1365-294x.2000.01020.x
Cun YZ, Wang XQ (2010) Plant recolonization in the Himalaya from the southeastern Qinghai-Tibetan Plateau: geographical isolation contributed to high population differentiation. Molec Phylogen Evol 56:72–982. https://doi.org/10.1016/j.ympev.2010.05.007
DeChaine EG, Martin AP (2005) Marked genetic divergence among sky island populations of Sedum lanceolatum (Crassulaceae) in the Rocky Mountains. Amer J Bot 92:477–486. https://doi.org/10.3732/ajb.92.3.477
Dlugosch KM, Parker IM (2008) Founding events in species invasions: genetic variation, adaptive evolution, and the role of multiple introductions. Molec Ecol 17:431–449. https://doi.org/10.1111/j.1365-294X.2007.03538.x
Drummond AJ, Suchard MA, Xie D, Rambaut A (2012) Bayesian phylogenetics with BEAUti and the BEAST 1.7. Molec Biol Evol 29:1969–1973. https://doi.org/10.1093/molbev/mss075
Eckert CG, Samis KE, Lougheed SC (2008) Genetic variation across species’ geographical ranges: the central–marginal hypothesis and beyond. Molec Ecol 17:1170–1188. https://doi.org/10.1111/2Fj.1365-294X.2007.03659.x
Eggli U (ed) (2005) Illustrated handbook of succulent plants: Crassulaceae. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-642-55874-0_2
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Excoffier L, Laval G, Schneider S (2005) Arlequin version 3.0: an integrated software package for population genetics data analysis. Evol Bioinform 1:47–50. https://doi.org/10.1177/117693430500100003
Fjeldsaå J, Lovett JC (1997) Geographical patterns of old and young species in African forest biota: the significance of specific montane areas as evolutionary centres. Biodivers Conservation 6:325–346. https://doi.org/10.1023/A:1018356506390
Fu PC, Gao QB, Zhang FQ, Xing R, Khan G, Wang JL, Liu H-R, Chen SL (2016) Responses of plants to changes in Qinghai-Tibetan Plateau and glaciations: evidence from phylogeography of a Sibiraea (Rosaceae) complex. Biochem Syst Ecol 65:72–82. https://doi.org/10.1016/j.bse.2016.01.006
Galtier N, Gouy M, Gautier C (1996) Seaview and phylo-win: two graphic tools for sequence alignment and molecular phylogeny. Comput Appl Biosci 12:543–548
Gao QB, Zhang DJ, Chen SY, Duan YZ, Zhang FQ, Li YH, Chen S (2009) Chloroplast DNA phylogeography of Rhodiola alsia (Crassulaceae) in the Qinghai-Tibet Plateau. Botany 87:1077–1088. https://doi.org/10.1139/B09-059
Gao QB, Zhang DJ, Duan YZ, Zhang FQ, Li YH, Fu PC, Chen S (2012) Intraspecific divergences of Rhodiola alsia (Crassulaceae) based on plastid DNA and internal transcribed spacer fragments. Bot J Linn Soc 168:204–215. https://doi.org/10.1111/j.1095-8339.2011.01193.x
Gao QB, Zhang FQ, Xing R, Gornall RJ, Fu PC, Li Y, Gengji ZM, Chen SL (2016) Phylogeographic study revealed microrefugia for an endemic species on the Qinghai-Tibetan Plateau: Rhodiola chrysanthemifolia (Crassulaceae). Pl Syst Evol 302:1179–1193. https://doi.org/10.1007/s00606-016-1324-4
Gontcharova SB (2006) Sedoideae, Crassulaceae of the Russian Far East flora. Dalnauka, Vladivostok (in Russian)
Gontcharova SB, Artyukova EV, Gontcharov AA (2006) Phylogenetic relationships among members of the subfamily Sedoideae (Crassulaceae) inferred from the ITS region sequences of nuclear rDNA. Russ J Genet 42:654–661. https://doi.org/10.1134/S102279540606010X
Gussarova G, Allen GA, Mikhaylova Y, McCormick LJ, Mirré V, Marr KL, Hebra RJ, Brochmann C (2015) Vicariance, long-distance dispersal, and regional extinction–recolonization dynamics explain the disjunct circumpolar distribution of the arctic-alpine plant Silene acaulis. Amer J Bot 102:1703–1720. https://doi.org/10.3732/ajb.1500072
Hais M, Komprdová K, Ermakov N, Chytrý M (2015) Modelling the last glacial maximum environments for a refugium of Pleistocene biota in the Russian Altai Mountains, Siberia. Palaeogeogr Palaeoclimatol Palaeoecol 438:135–145. https://doi.org/10.1016/j.palaeo.2015.07.037
Harpending HC, Batzer MA, Gurven M, Jorde LB, Rogers AR, Sherry ST (1998) Genetic traces of ancient demography. Proc Natl Acad Sci USA 95:1961–1967. https://doi.org/10.1073/pnas.95.4.1961
Hewitt GM (2000) The genetic legacy of the Quaternary ice ages. Nature 405:907–913. https://doi.org/10.1038/35016000
Hewitt GM (2004) Genetic consequences of climatic oscillations in the quaternary. Philos Trans Roy Soc London B Biol Sci 359:183–195. https://doi.org/10.1098/rstb.2003.1388
Hewitt GM (2011) Quaternary phylogeography: the roots of hybrid zones. Genetica 139:617–638. https://doi.org/10.1007/s10709-011-9547-3
Jump AS, Mátyás C, Peñuelas J (2009) The altitude-for-latitude disparity in the range retractions of woody species. Trends Ecol Evol 24:694–701. https://doi.org/10.1016/j.tree.2009.06.007
Kaltenrieder P, Belis CA, Hofstetter S, Ammann B, Ravazzi C, Tinner W (2009) Environmental and climatic conditions at a potential Glacial refugial site of tree species near the Southern Alpine glaciers. New insights from multiproxy sedimentary studies at Lago della Costa (Euganean Hills, Northeastern Italy). Quatern Sci Rev 28:2647–2662. https://doi.org/10.1016/j.quascirev.2009.05.025
Kelchner SA (2000) The evolution of non-coding chloroplast DNA and its application in plant systematics. Ann Missouri Bot Gard 87:499–527. https://doi.org/10.2307/2666142
Kienast F, Siegert C, Dereviagin A, Mai DH (2001) Climatic implications of Late Quaternary plant macrofossil assemblages from the Taymyr Peninsula, Siberia. Global Planet Change 31:265–281. https://doi.org/10.1016/S0921-8181(01)00124-2
Krasnoborov IM (1976) Alpine flora of the Western Sayan. Nauka, Novosibirsk (in Russian)
Lachmuth S, Durka W, Schurr FM (2010) The making of a rapid plant invader: genetic diversity and differentiation in the native and invaded range of Senecio inaequidens. Molec Ecol 19:3952–3967. https://doi.org/10.1111/j.1365-294X.2010.04797.x
Lehmkuhl F, Klinge M, Stauch G (2011) The extent and timing of late Pleistocene glaciations in the Altai and neighbouring mountain systems, 1st edn. In: Ehlers J, Gibbard PL, Hughes PD (eds) Quaternary glaciations - extent and chronology. A closer look, vol. 15. Elsevier, pp 967–979. https://doi.org/10.1016/B978-0-444-53447-7.00069-6
Levin DA, Samuel V (2017) On the young age of intraspecific herbaceous taxa. New Phytol 213:1513–1520. https://doi.org/10.1111/nph.14224
Li Z, Zou J, Mao K, Lin K, Li H, Liu J, Källman T, Lascoux M (2012) Population genetic evidence for complex evolutionary histories of four high altitude juniper species in the Qinghai-Tibetan Plateau. Evolution Int J Organic Evol 66:831–845. https://doi.org/10.1111/j.1558-5646.2011.01466.x
Li YC, Zhong DL, Rao GY, Wen J, Ren Y, Zhang JQ (2018) Gone with the trees: phylogeography of Rhodiola sect Trifida (Crassulaceae) reveals multiple refugia on the Qinghai-Tibetan plateau. Molec Phylogen Evol 121:110–120. https://doi.org/10.1016/j.ympev.2018.01.001
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Loveless MD, Hamrick JL (1984) Ecological determinants of genetic structure in plant populations. Annual Rev Ecol Evol Syst 15:65–95. https://doi.org/10.1146/annurev.es.15.110184.000433
Malyshev LI, Peshkova GA (1984) Osobennosti i genezis flory Sibiri (Predbaykale i Zabaykale). Nauka, Novosibirsk, p. 265 (in Russian)
Malyshev LI, Doronkin VM, Zuev VV, Baikov KS (2012) Conspectus Florae Rossiae Asiaticae: Plantae Vasculares. SB RAS, Novosibirsk (in Russian)
Mayuzumi S, Ohba H (2004) The phylogenetic position of East Asian Sedoideae (Crassulaceae) inferred from chloroplast and nuclear DNA sequences. Syst Bot 29:587–598. https://doi.org/10.1600/0363644041744329
Médail F, Diadema K (2009) Glacial refugia influence plant diversity patterns in the Mediterranean Basin. J Biogeogr 36:1333–1345. https://doi.org/10.1111/j.1365-2699.2008.02051.x
Mort ME, Soltis DE, Soltis PS, Francisco-Ortega J, Santos-Guerra A (2001) Phylogenetic relationships and evolution of Crassulaceae inferred from matK sequence data. Amer J Bot 88:76–91. https://doi.org/10.2307/2657129
Muellner-Riehl AN (2019) Mountains as evolutionary arenas: patterns, emerging approaches, paradigm shifts, and their implications for plant phylogeographic research in the Tibeto-Himalayan Region. Frontiers Pl Sci 10:195. https://doi.org/10.3389/fpls.2019.00195
Nagy LG, Kocsube S, Csanadi Z, Kovacs GM, Petkovits T et al (2012) Re-mind the gap! insertion: deletion data reveal neglected phylogenetic potential of the nuclear ribosomal internal transcribed spacer (ITS) of Fungi. PLoS ONE 7:49794. https://doi.org/10.1371/journal.pone.0049794
Nei M, Tajima F (1983) Maximum likelihood estimation of the number of nucleotide substitutions from restriction sites data. Genetics 105:207–217
Nikulin AY, Nikulin VY, Gonctharova SB, Gontcharov AA (2015a) ITS rDNAsequence comparisons resolve phylogenetic relationships in Orostachys subsection Appendiculatae. Pl Syst Evol 301:1441–1453. https://doi.org/10.1007/s00606-014-1165-y
Nikulin AY, Nikulin VY, Gontcharov AA (2015b) To the question of phylogenetic structure of the tribe Telephieae (Sempervivoideae, Crassulaceae) on the base of rDNA sequence comparisons. Bot Zhurn 100:1030–1040 (in Russian)
Ohba H (1978) Generic and infrageneric classification of the Old World Sedoideae (Crassulaceae). J Fac Sci Univ Tokyo III 12:139–198
Ohba H (1987) Biogeography of the genus Rhodiola. Acta Phytotax Geobot 38:211–223. https://doi.org/10.18942/bunruichiri.KJ00002992258
Ono Y, Shulmeister J, Lehmkuhl F, Asahi K, Aoki T (2004) Timings and causes of glacial advances across the PEP-II transect (East-Asia to Antarctica) during the last glaciation cycle. Quatern Int 118:55–68. https://doi.org/10.1016/S1040-6182(03)00130-7
Opgenoorth L, Vendramin GG, Mao K, Miehe G, Miehe S, Liepelt S, Liu J, Ziegenhagen B (2010) Tree abundance on the Tibetan Plateau marks the world’s highest known tree line of the Last Glacial Maximum. New Phytol 185:332–342. https://doi.org/10.1111/j.1469-8137.2009.03007.x
Palstra FP, Ruzzante DE (2008) Genetic estimates of contemporary effective population size: what can they tell us about the importance of genetic stochasticity for wild population persistence? Molec Ecol 17:3428–3447. https://doi.org/10.1111/j.1365-294X.2008.03842.x
Petit RJ, Aguinagalde I, De Beaulieu J-L, Bittkau C, Brewer S, Cheddadi R, Ennos R, Fineschi S, Grivet D, Lascoux M, Mohanty A, Müller-Starck G, Demesure-Musch B, Palme A, Martin JP, Rendell S, Vendramin GG (2003) Glacial refugia: hotspots but not melting pots of genetic diversity. Science 300:1563–1565. https://doi.org/10.1126/science.1083264
Pons O, Petit RJ (1996) Measuring and testing genetic differentiation with ordered versus unordered alleles. Genetics 144:1237–1245
Qiu Y, Liu Y, Kang M, Yi G, Huang H (2013) Spatial and temporal population genetic variation and structure of Nothotsuga longibracteata (Pinaceae), a relic conifer species endemic to subtropical China. Genet Molec Biol 36:598–607. https://doi.org/10.1590/S1415-47572013000400019
Rambaut A (2009) FigTree version 1.3.1. Available at: http://tree.bio.ed.ac.uk/software/figtree. Accessed 26 Jun 2017
Rambaut A, Drummond AJ (2007) Tracer v1.4: MCMC trace analyses tool. Available at: http://beast.bio.ed.ac.uk/Tracer. Accessed 26 Jun 2017
Revushkin AS (1988) The alpine flora of the Altai. Tomsk University Press, Tomsk (in Russian)
Řičánková VP, Robovský J, Riegert J (2014) Ecological structure of recent and last glacial mammalian faunas in northern Eurasia: the case of Altai-Sayan refugium. PLoS ONE 9:e85056. https://doi.org/10.1371/journal.pone.0085056
Rogers AR, Harpending H (1992) Population growth makes waves in the distribution of pairwise differences. Molec Biol Evol 9:552–569. https://doi.org/10.1093/oxfordjournals.molbev.a040727
Roman J, Darling JA (2007) Paradox lost: genetic diversity and the success of aquatic invasions. Trends Ecol Evol 22:454–464. https://doi.org/10.1016/j.tree.2007.07.002
Ronikier M, Cieślak E, Korbecka G (2008) High genetic differentiation in the alpine plant Campanula alpina Jacq. (Campanulaceae): evidence for glacial survival in several Carpathian regions and long-term isolation between the Carpathians and the Alps. Molec Ecol 17:1763–1775. https://doi.org/10.1111/j.1365-294X.2008.03664.x
Schmitt T, Varga Z (2012) Extra-Mediterranean refugia: the rule and not the exception? Frontiers Zool 9:22. https://doi.org/10.1186/1742-9994-9-22
Shaw J, Lickey EB, Beck JT, Farmer SB, Wusheng L, Miller J, Siripun KC, Winder CT, Schilling EE, Small RL (2005) The tortoise and the hare II: relative utility of 21 noncoding chloroplast DNA sequences for phylogenetic analysis. Amer J Bot 92:142–166. https://doi.org/10.3732/ajb.92.1.142
Shaw J, Lickey EB, Schilling EE, Small RL (2007) The Tortoise and the Hare III. Comparison of whole chloroplast genome sequences to choose noncoding regions for phylogenetic studies in angiosperms. Amer J Bot 94:275–288. https://doi.org/10.3732/ajb.94.3.275
Stehlik I, Schneller JJ, Bachmann K (2002) Immigration and in situ glacial survival of the low-alpine Erinus alpinus (Scrophulariaceae). Biol J Linn Soc 77:87–103. https://doi.org/10.1046/j.1095-8312.2002.00094.x
Stepanov NV, Vasiliev AN, Tupitcina NN, Antipova EM, Sonnikova AE, Andreeva EB, Shtarker VV, Belik OV, Matveeva YuV (2003) Flora of Sayan. Krasnoyarsk University Press, Krasnoyarsk (in Russian)
Sun XJ, Chen YS (1991) Palynological records of the last 11,000 years in China. Quatern Sci Rev 10:537–544. https://doi.org/10.1016/0277-3791(91)90047-X
Taberlet P (1998) Biodiversity at the intraspecific level: the comparative phylogeographic approach. J Biotechnol 64:91–100. https://doi.org/10.1016/S0168-1656(98)00106-0
Taberlet P, Fumagalli L, Wust-Saucy AG, Cosson JF (1998) Comparative phylogeography and postglacial colonization routes in Europe. Molec Ecol 7:453–464. https://doi.org/10.1046/j.1365-294x.1998.00289.x
Takhtajan A (1986) Floristic regions of the world. University of California Press, Berkeley
Tribsch A, Schönswetter P (2003) Patterns of endemism and comparative phylogeography confirm palaeoenvironmental evidence for Pleistocene refugia in the Eastern Alps. Taxon 52:477–497. https://doi.org/10.2307/3647447
Tzedakis P, Lawson I, Frogley M, Hewitt G, Preece R (2002) Buffered tree population changes in a Quaternary refugium: evolutionary implications. Science 297:2044–2047. https://doi.org/10.1126/science.1073083
Uller T, Leimu R (2011) Founder events predict changes in genetic diversity during human-mediated range expansions. Global Change Biol 17:3478–3485. https://doi.org/10.1111/j.1365-2486.2011.02509.x
Wen J, Zhang J, Nie ZL, Zhong Y, Sun H (2014) Evolutionary diversifications of plants on the Qinghai-Tibetan Plateau. Frontiers Genet 5:4. https://doi.org/10.3389/fgene.2014.00004
Wen J, Nie ZL, Ickert-Bond SM (2016) Intercontinental disjunctions between eastern Asia and western North America in vascular plants highlight the biogeographic importance of the Bering land bridge from late Cretaceous to Neogene. J Syst Evol 54:469–490. https://doi.org/10.1111/jse.12222
Wolfe KH, Li WH, Sharp PM (1987) Rates of nucleotide substitution vary greatly among plant mitochondrial, chloroplast, and nuclear DNAs. Proc Natl Acad Sci USA 84:9054–9058. https://doi.org/10.1073/pnas.84.24.9054
Wroblewska A (2012) The role of disjunction and postglacial population expansion on phylogeographical history and genetic diversity of the circumboreal plant Chamaedaphne calyculata. Biol J Linn Soc 105:761–775. https://doi.org/10.1111/j.1095-8312.2011.01828.x
Xu T, Abbott RJ, Milne RI, Mao K, Du FK, Wu G, Ciren Z, Miehe G, Liu J (2010) Phylogeography and allopatric divergence of cypress species (Cupressus L.) in the Qinghai-Tibetan Plateau and adjacent regions. BMC Evol Biol 10:194. https://doi.org/10.1186/1471-2148-10-194
Yamane K, Yano K, Kawahara T (2006) Pattern and rate of INDEL evolution inferred from whole chloroplast intergenic regions in sugarcane, maize and rice. DNA Res 13:197–204. https://doi.org/10.1093/dnares/dsl012
Zhang YH, Volis S, Sun H (2010) Chloroplast phylogeny and phylogeography of Stellera chamaejasme on the Qinghai-Tibet Plateau and in adjacent regions. Molec Phylogen Evol 57:1162–1172. https://doi.org/10.1016/j.ympev.2010.08.033
Zhang JQ, Meng SY, Rao GY (2014a) Phylogeography of Rhodiola kirilowii (Crassulaceae): a story of miocene divergence and quaternary expansion. PLoS ONE 9:e112923. https://doi.org/10.1371/journal.pone.0112923
Zhang JQ, Meng SY, Wen J, Rao GY (2014b) Phylogenetic relationships and character evolution of Rhodiola (Crassulaceae) based on nuclear ribosomal ITS and plastid trnL–F and psbA–trnH sequences. Syst Bot 39:441–451. https://doi.org/10.1600/036364414X680753
Zhang YZ, Zhu RW, Zhong DL, Zhang JQ (2018) Nunataks or massif de refuge? A phylogeographic study of Rhodiola crenulata (Crassulaceae) on the world’s highest sky islands. BMC Evol Biol 18:154. https://doi.org/10.1186/s12862-018-1270-6
Acknowledgements
We thank Shamil Abdullin, Vadim Bakalin, Viktor Bogatov, Roman Dudkin, Konstantin Kiselev, Alexandra Dubrovina, Yuri Ovchinnikov, Valery Shokhrin, Valentina Shokhrina, Dmitry Sidorov and Valentin Yakubov for sampling natural populations of O. spinosa; Marko Dobos for sharing his personal Orostachys collection and Sun Yan for assistance during sampling in North-Eastern China.
Funding
The study was supported by Russian Foundation for Basic Research according to the research Project No. 18-34-00436.
Author information
Authors and Affiliations
Contributions
AN and AG conceived the paper with VN. AN performed the analyses and wrote the manuscript under the guidance of AG and with critical reviews, editing and contributions from VN.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Mike Thiv.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online Resource 1. GenBank accession numbers of the cpDNA sequences.
Online Resource 2. Alignment used in analyses.
Online Resource 3. Bayesian chronogram of cladogenesis of Orostachys spinosa populations.
Rights and permissions
About this article
Cite this article
Nikulin, A.Y., Nikulin, V.Y. & Gontcharov, A.A. Orostachys spinosa (Crassulaceae) origin and diversification: East Asia or South Siberian Mountains? Chloroplast DNA data. Plant Syst Evol 306, 84 (2020). https://doi.org/10.1007/s00606-020-01713-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-020-01713-4