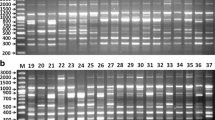
Abstract
Evolution of genus is accompanied by ecological diversification. The majority of species grow in open, sunny, rather dry sites in arid and moderately humid climates. However, Allium species have adapted for many other ecological niches. Classical approaches for the identification of Allium cultivars are based on morphological traits. The assessment of these traits is difficult and their evaluation can be subjective considering that most of these cultivars are closely related. Hence, this study of Internal Transcribed Spacer (ITS) sequencing and four barcoding regions, matK, rbcL, trnH-psbA, trnL and Inter Simple Sequence Repeats (ISSR) were researched in onion, Allium cepa L. (Alliaceae) collected from three different cultivation sites. The results established noticeable hereditary divergence among the three cultivars. In ITS and matK, BDUT 1453, BDUT 1454 and BDUT 1455 were independent of each other and formed three clusters. In rbcL, BDUT 1453 formed an independent cluster from the cluster of BDUT 1454 and BDUT 1455. But in trnH-psbA, BDUT 1454 formed an independent cluster and BDUT 1453 and BDUT 1455 were closely placed whereas trnL showed all the three forming a cluster wherein BDUT 1453 and BDUT 1454 were placed closely in a sub-cluster. In ISSR, BDUT 1454 and BDUT 1455 formed a single cluster and BDUT 1453 diverged from it. Even though the tested cultivars belong to the same species they showed genetic divergence among themselves.













Similar content being viewed by others
Data accessibility
Genebank Accession numbers: KF769488, KF769489, KF769490, KF769494, KF769495, KF769496, KF769500, KF769501, KF769502, KF769506, KF769507, KF769508, KF779156, KF779157, KF779156.
References
Anvarkhah S, Khajeh MH, Hassan Rashed MM, Ali Davari EP, Hashemi H (2013) Identification of three species of genus Allium using DNA barcoding. Intl J Agri Crop Sci 5:1195–1203
Azizi A, Wagner C, Honermeier B, Wolfgang F (2009) Intraspecific diversity and relationship between subspecies of Origanum vulgare revealed by comparative AFLP and SAMPL marker analysis. Plant Syst Evol 281:151–160. https://doi.org/10.1007/s00606-009-0197-1
Bafeel SO, Arif IA, Bakir MA, Khan HA, Farhan AHA, Homaidan AAA, Ahamed A, Thomas J (2011) Comparative evaluation of PCR success with universal primers of maturase K (matK) and ribulose-1,5-bisphosphate carboxylase/oxygenase large subunit (rbcL) for barcoding of some arid plants. Plant Omics 4:195–198
Baldwin S, Pither-Joyce M, Wright K, Chen L, McCallum J (2012) Development of robust genomic simple sequence repeat markers for estimation of genetic diversity within and among bulb onion (Allium Cepa L.) populations. Molecular Breeding 30:1401–1411. https://doi.org/10.1007/s11032-012-9727-6
Baldwin BG, Sanderson MJ, Porter JM, Wojciechowski MF, Campbell CS, Donoghue MJ (1995) The ITS region of nuclear ribosomal DNA: a valuable source of evidence on angiosperm phylogeny. Ann Missouri Bot Gar 82:247–277. https://doi.org/10.2307/2399880
Becker J, Heun M (1994) Microsatellites: allele variation and mapping. Plant Molr Biol 27:835–845
Bhasi A, Senalik D, Simon PW, Kumar B, Manikandan V, Philip P, Senapathy P (2010) RoBust: an integrated genomics resource for the root and bulb crop families Apiaceae and Alliaceae. BMC Plant Biol 10:161. https://doi.org/10.1186/1471-2229-10-161
Bianco CL, Fernandez JA, Migliaro D, Crino P, Egea-Gilabert C (2011) Identification of F1 hybrids of artichoke by ISSR markers and morphological analysis. Mol Breed 27:157–170. https://doi.org/10.1007/s11032-010-9418-0
Blattner FR, Kadereit JW (1999) Morphological evolution and ecological diversification of the forest dwelling poppies (papaveracea: Chelidonioideae) as deduced from a molecular phylogeny of the ITS region. Plant Syst Evol 219:181–197. https://doi.org/10.1007/BF00985578
Brooks HJ, Vest G (1985) Public programs on genetics and breeding of horticultural crops in the United States. Hort Sci 20:826–830. https://doi.org/10.21273/HORTSCI.20.5.826
CBOL Plant Working Group (2009) A DNA barcode for land plants. Proc Natl Acad Sci USA 106:12794–12797. https://doi.org/10.1073/pnas.090584510
Chalbi A, Chikh-Rouhou H, Mezghani N, Slim A, Fayos O, Bel-Kadhi MS, Garcés-Claver A (2023) Genetic diversity analysis of onion (Allium cepa L.) from the Arid region of Tunisia using phenotypic traits and SSR markers. Horticulturae 9(10):1098. https://doi.org/10.3390/horticulturae9101098
Chase MW, Cowan RS, Hollingsworth PM, van den Berg C, Madrinan S et al (2007) A proposal for a standardised protocol to barcode all land plants. Taxon 56:295–299. https://doi.org/10.1002/tax.562004
Chase MW, Fay MF (2009) Barcoding of plants and Fungi. Science 325:682–683
Chen S, Yao H, Han J, Liu C, Song J et al (2010) Validation of the ITS2 region as a novel DNA barcode for identifying medicinal plant species. PLoS ONE. https://doi.org/10.1371/journal.pone.0008613
Costion C, Ford A, Cross H, Crayn D, Harrington M, Lowe A (2011) Plant DNA barcodes can accurately estimate species richness in poorly known Floras. PLoS ONE. https://doi.org/10.1371/journal.pone.0026841
Cowan RS, Chase MW, Kress WJ, Savolainen V (2006) 300,000 species to identify: problems, progress, and prospects in DNA barcoding of land plants. Taxon 55:611–616. https://doi.org/10.2307/25065638
Da-Cruz F (2012) Sistemática e filogenética molecular do gênero Hexachlamys (Myrtaceae) através do uso de marcadores plastidiais e nucleares. Programa de Pos-Graduacaoem genetic e Biologia Molecular department de Genetica, Instituto de Bicietica, Universidade Federal do Rio Grande do Sul. http://hdl.handle.net/10183/49275
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Dubouzet JG, Shinoda K (1998) Phylogeny of Allium L. Subgenus Melanocrommyum (Webb Et Berth.) Rouy based on DNA sequence analysis of the internal transcribed spacer region of rDNA. Theor Appl Genet 97:541–549. https://doi.org/10.1007/s001220050929
Dubouzet JG, Shinoda K (1999) Relationships among old and new world Alliums according to ITS DNA sequence analysis. Theor Appl Genet 98:422–433. https://doi.org/10.1007/s001220051088
Fazekas AJ, Burgess KS, Kesanakurti PR et al (2008) Multiple multilocus DNA barcodes from the plastid genome discriminate plant species equally well. PLoS ONE. https://doi.org/10.1371/journal.pone.0002802
Frey KJ (1999) National plant breeding study-I. Special report 98. Iowa State University, Ames
Friesen N, Fritsch RM, Pollner S, Blattner FR (2000) Molecular and morphological evidence for an origin of the aberrant genus milula within himalayan species of Allium (Alliaceae). Mol Phylogenet Evol 17:209–218. https://doi.org/10.1006/mpev.2000.0844
Friesen N, Klaas M (1998) Origin of some minor vegetatively propagated Allium crops studied with RAPD and GISH. Genet Resour Crop Evol 45:511–523. https://doi.org/10.1023/A:1008647700251
Fritsch RM, Friesen N (2002) Evolution, domestication and taxonomy. In: Rabinowitch HD, Currah L (eds) Allium crop science: recenttt advances. CABI Publishing, New York, pp 5–30
Fu YM, Jiang WM, Fu CX (2011) Identification of species within Tetrastigma (Miq.) Planch, (Vitaceae) based on DNA barcoding techniques. J Syst Evol 49:237–245. https://doi.org/10.1111/j.1759-6831.2011.00126.x
Fujito S, Akyol TY, Mukae T, Wako T, Yamashita KI, Tsukazaki H, Hirakawa H, Tanaka K, Mine Y, Sato S, Shigyo M (2021) Construction of a high-density linkage map and graphical representation of the arrangement of transcriptome-based unigene markers on the chromosomes of onion, Allium cepa L. BMC Genomics 22(1):1–8. https://doi.org/10.1186/s12864-021-07803-y
Gao T, Yao H, Song J, Liu C, Zhu Y et al (2010) Identification of medicinal plants in the family Fabaceae using a potential DNA barcode ITS2. J Ethno Pharmacol 130:116–121. https://doi.org/10.1016/j.jep.2010.04.026
Goldman JJ (2008) The use of ISSR markers to identify Texas bluegrass inter specific hybrids. Plant Breed 127:644–646. https://doi.org/10.1111/j.1439-0523.2008.01526.x
Gostimsky SA, Kokaeva ZG, Konovlac FA (2005) Studying plant genome variation using molecular markers. Russ Genet 41:378–388. https://doi.org/10.1007/s11177-005-0101-1
Gounaris Y, Skoula M, Fournaraki C, Drakakaki G, Makris A (2002) Comparison of essential oils and genetic relationship of Origanumx intercedents to its parental taxa in the island of Crete. Biochem Syst Ecol 30:249–258. https://doi.org/10.1016/S0305-1978(01)00079-5
Gurcharan SS (2004) Plant systematics: an integrated approach. Science publishers, Enfield, NH
Hajibabaei M, Singer GAC, Hebert PDN, Hickey DA (2007) DNA barcoding: how it complements taxonomy, molecular phylogenetics and population genetics. Trends Genet 23:167–172. https://doi.org/10.1016/j.tig.2007.02.001
Hao G, Lee DH, Lee JS, Lee NS (2002) A study of taxonomical relationships among species of Korean Allium sect. Sacculiferum (Alliaceae) and related species using inter-simple sequence repeat (ISSR) markers. Bot Bull Acad Sin 43:63–68
Hayden MJ, Tabone TL, Nguyen TMS, Keiper FJ, Fox RL, Chalmers KJ, Eglinton JA (2010) An informative set of SNP markers for molecular characterization of Australian barley germplasm. Crop past Sci 61:70–83. https://doi.org/10.1071/CP09140
Hebert PDN, Cywinska A, Ball SL, DeWaard JR (2003) Biological identification through DNA barcodes. Proc R Soc Lond Series B 270:313–321. https://doi.org/10.1098/rspb.2002.2218
Herlina L, Reflinur R, Sobir S, Maharijaya A, Wiyono S (2019) The genetic diversity and population structure of shallots (Allium cepa var. aggregatum) in Indonesia based on R gene-derived markers. Biodiversitas J Biol Divers 20(3):696–703. https://doi.org/10.13057/biodiv/d200312
Hirschegger P, Jakse J, Tronteli P, Bohanec B (2010) Origins of Allium ampeloprasum horticultural groups and a molecular phylogeny of the section Allium (Allium: Alliaceae). Mol Phylogenet Evol 54:488–497. https://doi.org/10.1016/j.ympev.2009.08.030
Hollingsworth PM, Forrest LL, Spouge JL, Hajibabaei M, Ratnasingham S et al (2009) Refining the DNA barcode for land plants. Proc Natl Acad Sci USA 106:12794–12797. https://doi.org/10.1073/pnas.111681210
Hsiao C, Jacobs SWL, Chatterton NJ, Asay KH (1999) A molecular phylogeny of the grass family (Poaceae) based on the sequence of nuclear ribosomal DNA (ITS). Aust Syst Bot 11:667–688. https://doi.org/10.1071/SB97012
Ipek M, Ipek A, Simon PW (2008) Genetic characterization of Allium tuncelianum: an endemic edible Allium species with garlic odor. Sci Hortic 115:409–415. https://doi.org/10.1016/j.scienta.2007.11.002
Jabbes N, Geoffriau E, Clerc VL, Dridi B, Hannechi C (2011) Inter simple sequence repeat fingerprints for assess genetic diversity of Tunisian garlic populations. J Agri Sci 3:77–85
Jayaswall K, Sagar R, Jayaswal D, Kumar A, Singh SP, Seth R, Sharma H, Kumar D, Mahajan V, Kumar S, Singh M (2024) Development of Allium cepa potential intron polymorphism markers for molecular breeding of alliums. South Afr J Bot 164:209–220. https://doi.org/10.1016/j.sajb.2023.11.050
Jayaswall K, Sharma H, Bhandawat A, Sagar R, Jayaswal D, Kumar A, Chaturvedi P, Mahajan V, Kumar S, Singh M (2022) Chloroplast derived SSRs reveals genetic relationships in domesticated alliums and wild relatives. Genet Resour Crop Evol 69:363–372. https://doi.org/10.1007/s10722-021-01235-z
Jayaswall K, Sharma H, Bhandawat A, Sagar R, Yadav VK, Sharma V, Mahajan V, Roy J, Singh M (2019) Development of intron length polymorphic (ILP) markers in onion (Allium cepa L.), and their cross-species transferability in garlic (A. sativum L.) and wild relatives. Genet Resour Crop Evol 66:1379–1388. https://doi.org/10.1007/s10722-019-00808-3
Khade YP, Salunkhe SR, Manjunathagowda DC, Sinhasane SR, Mahidar Gowd TY, Mahajan V, Singh M (2022) Molecular characterization of short-day onion genotypes by intron length polymorphic (ILP) markers. Genet Resour Crop Evol 69:2077–2086. https://doi.org/10.1007/s10722-022-01398-3
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/BF01731581
Kress WJ, Erickson DL (2007) A two locus global DNA barcode for land plants: the coding rbcLgene complements the non-coding trnh-psba spacer region. PLoS ONE. https://doi.org/10.1371/journal.pone.0000508
Kress WJ, Wurdack KJ, Zimmer EA, Weigt LA, Janzen DH (2005) Use of DNA barcodes to identify flowering plants. Proc Natl Acad Sci USA 102:8369–8374. https://doi.org/10.1073/pnas.050312310
Labra M, Miele M, Ledda B, Grassi F, Mazzei M, Sala F (2004) Morphological characterization, essential oil composition and DNA genotyping of Ocimum basilicum L. cultivars. Plant Sci 167:725–731. https://doi.org/10.1016/j.plantsci.2004.04.026
Lahaye R, van der Bank M, Bogarin D, Warner J, Pupulin F, Gigot G, Maurin O, Duthoit S, Barraclough TG, Savolainen V (2008) DNA barcoding the floras of biodiversity hotspots. Proc Natl Acad Sci USA 105:2923–2928. https://doi.org/10.1073/pnas.0709936105
Lewontin RC (1972) Testing the theory of natural selection. Nature 236:181–182
Liu Y, Yan HF, Cao T, Ge XJ (2010) Evaluation of 10 plant barcodes in Bryophyta (Mosses). J Syst Evol 48:38–46. https://doi.org/10.1111/j.1759-6831.2009.00063.x
Mallor C, Arnedo-Andrés MS, Garcés-Claver A (2014) Assessing the genetic diversity of Spanish Allium cepa landraces for onion breeding using microsatellite markers. Sci Hort 170:24–31. https://doi.org/10.1016/j.scienta.2014.02.040
McCallum J, Leite D, Pither-Joyce M, Havey MJ (2001) Expressed sequence markers for genetic analysis of bulb onion (Allium cepa L.). Theoretical and Applied Genetics 103:979–991. https://doi.org/10.1007/s001220100630.103
Mes THM, Fritsch RM, Pollner S, Bachmann K (1999) Evolution of the chloroplast genome and polymorphic ITS regions in Allium subg. Melanocrommyum. Genome 42:237–247. https://doi.org/10.1139/g98-123
Moon JS, Kim B, Kim S (2023) Evaluation of homozygosity levels of onion (Allium cepa L.) inbred lines using molecular markers evenly distributed in a high-resolution linkage map. Euphytica 219(9):96. https://doi.org/10.1007/s10681-023-03226-w
Mukherjee A, Sikhar B, Ghosh B, Banerjee A, Ghosh E, Bhattacharya M, Roy SC (2013) RAPD and ISSR analysis of some economically important species, varieties, and cultivars of the genus Allium (Alliaceae). Turk J Bot 37:605–618. https://doi.org/10.3906/bot-1208-18
Nagaoka T, Ogihara Y (1997) Applicabillity of inter-simple sequence repeat polymorphisms in wheat for use as DNA markers in comparison to RFLP and RAPD markers. Theor Appl Genet 94:597–602. https://doi.org/10.1007/s001220050456
Nei M (1972) Genetic distance between populations. Amer Naturalist 106:283–292. https://doi.org/10.1086/282771
Newmaster SG, Fazekas AJ, Ragupathy S (2006) DNA barcoding in land plants: evaluation of rbcL in a multigene tiered approach. Can J Bot 84:335–341. https://doi.org/10.1139/b06-047
Newmaster SG, Fazekas AJ, Steeves RAD, Janovec J (2008) Testing candidate plant barcode regions in the Myrisiticaceae. Mol Ecol Notes 8:480–490. https://doi.org/10.1111/j.1471-8286.2007.02002.x
Nguyen NH, Driscoll HE, Specht CD (2008) A molecular phylogeny of the wild onions (Allium; Alliaceae) with a focus on the western north American center of diversity. Mol Phylogenet Evol 47:1157–1172. https://doi.org/10.1016/j.ympev.2007.12.006
Novak J, Lukas B, Bolzer K, Grausgruber-Groger S, Dengenhardt J (2008) Identification and characterization of simple sequence repeat markers from a glandular Origanum vulgare expressed sequence tag. Mol Ecol Notes 8:599–601. https://doi.org/10.1111/j.1471-8286.2007.02059.x
Noyes RD, Rieseberg LH (1998) ITS sequence data support a single origin for north American Astereae (Asteraceae) and reflect deep geographic divisions asters. S.l. Am J Bot 86:398–412. https://doi.org/10.2307/2656761
Pathirana R, Carimi F (2022) Management and utilization of plant genetic resources for a sustainable agriculture. Plants. https://doi.org/10.3390/plants11152038
Peng X, Liu JJ, Xiang Y, Huang S (2006) A practical handbook of plant molecular biotechnology. Chemical Industry Press, Beijing
Poczai P, Matyas KK, Szabo I, Varga I, Hyvonen J, Cernak I, Gorji AM, Decsi K, Taller J (2011) Genetic variability of Thermal Nymphaea (Nymphaeaceae) population based on ISSR markers: implications on relationships, hybridization, and conservation. Plant Mol Biol Rep 29:906–918. https://doi.org/10.1007/s11105-011-0302-9
Raj AC, Sharangi AB, Das A, Pramanik K, Upadhyay TK, Almutairi M, Khan MI, Ahmad I, Kausar MA, Saeed M (2022) Assessing the genetic divergence of onion (Allium Cepa L.) through morpho-physiological and molecular markers. Sustainability 14(3):1131. https://doi.org/10.3390/su14031131
Reddy PM, Sarla N, Siddiq EA (2002) Inter simple sequence repear (SSR) polymorphism and its application in plant breeding. Euphytica 128:9–17. https://doi.org/10.1023/A:1020691618797
Rizkalla AA, Attia SAA, El-Hady AAA, Hanna NS, Nasseef JE (2012) Genetic diversity based on ISSR and protein markers associated with Earliness Trait in Wheat. World Appl Sci J 20:23–33
Schori M, Schowalter AM (2011) DNA barcoding as a means for identifying Medicinal plants of Pakistan. Pak J Bot 43:1–4
Segarra-Moragues JG, Gleiser G (2009) Isolation and characterisation of di and tri nucleotide microsatellite loci in Rosmarinus officinalis (Lamiaceae), using enriched genomic libraries. Conserv Genet 10:571–575. https://doi.org/10.1007/s10592-008-9572-7
Singh PR, Mahajan V, Verma A, Sagar R, Jayaswall K, Shukla N, Gedam P, Singh M (2021) Identification and characterization of white onion (Allium cepa L.) genotypes for high total soluble solid content through molecular markers. Genet Resour Crop Evol 68:957–967. https://doi.org/10.1007/s10722-020-01037-9
Sonnante G, Pignone D (2001) Assessment of genetic variation in a collection of Lentil using molecular tools. Euphytica 120:301–307. https://doi.org/10.1023/A:1017568824786
Taberlet P, Coissac E, Pompanon F, Gielly L, Miquel C, Valentini A (2007) Power and limitations of the chloroplast trnL (UAA) intron for plant DNA barcoding. Nucleic Acids Res. https://doi.org/10.1093/nar/gkl938
Tamura K, Nei M (1993) Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol Biol Evol 10:512–526. https://doi.org/10.1093/oxfordjournals.molbev.a040023
Tautz D (1989) Hypervariability of simple sequence as a general source: a polymorphic DNA markers. Nucleic Acids Res 17:6463–6471
Tibayrenc M (2005) Bridging the gap between molecular epidemiologists and evolutionists. Trends Microbiol 13:575–580. https://doi.org/10.1016/j.tim.2005.09.004
Trindade H (2007) Molecular biology of aromatic plants and spices. Flavour Frag J 25:272–281. https://doi.org/10.1002/ffj.1974
Vijayan K, Tsou CH (2010) DNA barcoding in plants: taxonomy in a new perspective. Curr Sci 11:1530–1541
Vos P, Hogers R, Bleeker M, Reijans M, Van Der T, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23:4407–4414. https://doi.org/10.1093/nar/23.21.4407
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetids. In: Innis MA, Gelfand D, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, New York, pp 315–322
Williams JG, Kubelik AR, Livak KJ, Rafalski JA, Tingey SV (1990) DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucleic Acids Res 18:6531–6535. https://doi.org/10.1093/nar/18.22.6531
Zarei H, Fakheri BA, Naghavi MR, Mahdinezhad N (2020) Phylogenetic relationships of Iranian Allium species using the matK (cpDNA gene) region. J Plant Biotechnol 31:15–25. https://doi.org/10.5010/JPB.2020.47.1.015
Zietkiewciz E, Rafalksi A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-Anchored polymerase chain reaction amplification. Genomics 20:176–183. https://doi.org/10.1006/geno.1994.1151
Acknowledgements
The first author gratefully acknowledges Dr. M.B. Viswanathan, former Professor and Head, Department of Botany, Bharathidasan University, for given the lab facility for DNA and PCR analyses and all the authors are gratefully acknowledged Mr. R. Anand for his assistance in sample collection.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All the authors were equally contributed towards the conception and design of the project. N.C.J.P.L and R.R produced, assembled and analysed the data. The manuscript was written by N.C.J.P.L and D.M with valuable contributions from R.R. and J.R.B. All authors were reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
No approval is required for the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Packia Lekshmi, N.C.J., Brindha, J.R., Mahamuni, D. et al. Genomic profiling reveals intraspecific divergence and interspecific diversity in Allium cepa L.. Genet Resour Crop Evol (2024). https://doi.org/10.1007/s10722-024-01870-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10722-024-01870-2